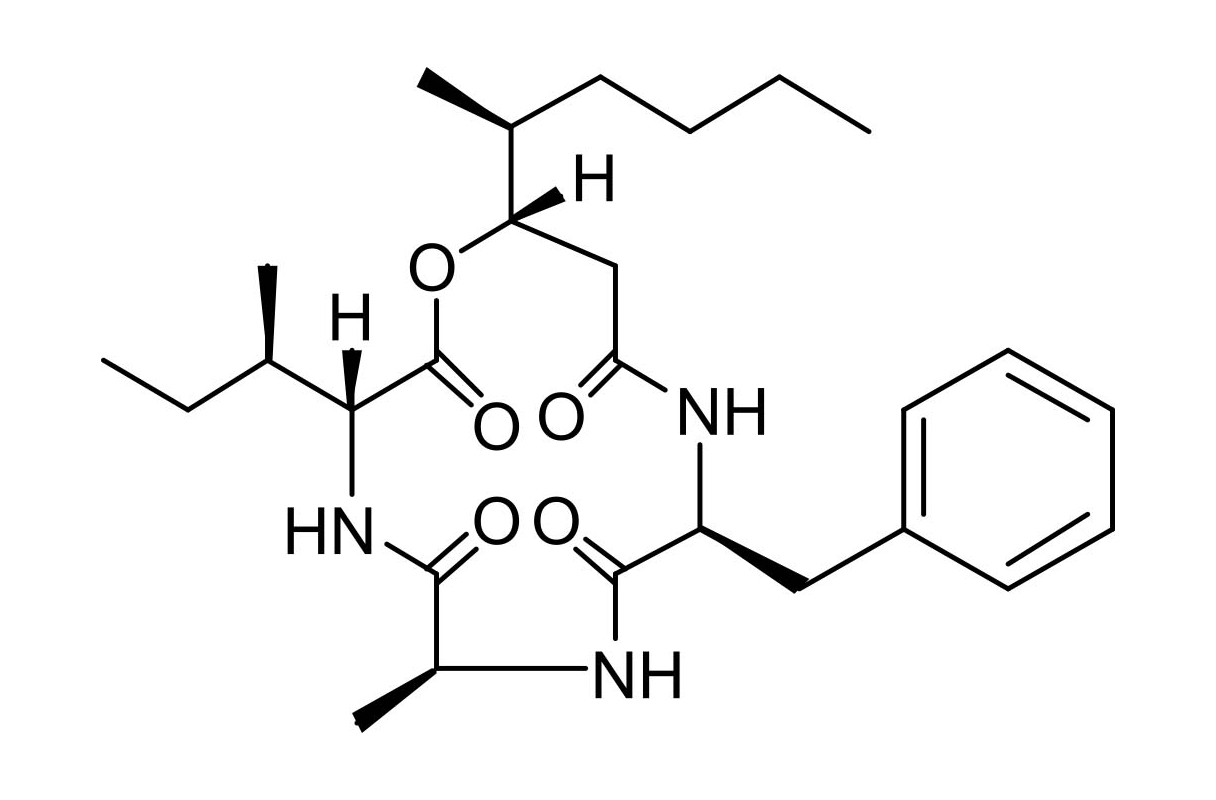
Beauveriolide III
Application Notes
Beauveriolides are 13-membered cyclodepsipeptides isolated from Beauveria sp. Beauveriolide III inhibits acyl-CoA:cholesterol acyltransferase (ACAT) to block the synthesis of cholesteryl ester, leading to a reduction of lipid droplets in macrophages. Beauverolide III is more potent against ACAT1 than ACAT2 (IC50 5.5 vs. > 20 µM, respectively). Beauveriolide III is antiatherogenic activity in mouse models.
References
- Absolute stereochemistry of fungal beauveriolide III and ACAT inhibitory activity of four stereoisomers. Ohshiro T. et al. J Org Chem. 2006, 71, 7643.
- Discovery and combinatorial synthesis of fungal metabolites beauveriolides, novel antiatherosclerotic agents. Tomoda H. and Doi T. Acc Chem Res 2008, 41, 32.
- The selectivity of beauveriolide derivatives in inhibition toward the two isozymes of acyl-CoA : cholesterol acyltransferase. Oshiro T. et al. Chem Pharm Bull. 2009, 57, 377.